A page in which David Pilling investigates the temperatures of the containers bulbs live in.
The temperature of bulbs in pots is of concern for a number of reasons. Many bulbs are destroyed by temperatures below freezing 0 °C turning them to 'mush'. Bulbs can also be destroyed by high temperatures, being literally 'cooked'. In between those two extremes, temperature may control when bulbs start to grow, when they become dormant, development during apparent dormancy, and flower production.
There are a number of often repeated statements about the temperatures of bulbs in pots. Using cheap and easily available temperature logging devices, some of these were experimentally tested.
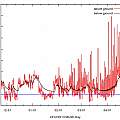
Bulbs in pots will get colder than bulbs planted in the ground (in Winter)
In the Winter of 2012/2013 temperature recorders were placed above and below ground (see the section at the end for details). The first graph shows the results. Temperatures 8 inches below ground were never less than 0 °C, those above ground remained below 0 °C for periods in excess of 24 hours, plenty of time to kill the bulbs of many species. The second graph shows the coldest days of the period. Graph 3 is of the 24 hour average temperature.
Download data
The theory is found to be correct, bulbs in the ground are warmer than those in pots. It is also evident that temperatures in the ground are more stable. Planting in the ground keeps bulbs cooler too, this may matter if you are growing species where high temperatures trigger dormancy.
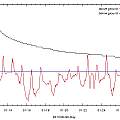
Bulbs in pots left in the sun can get too hot
To test this idea one temperature logger was placed in an 8 inch pot of compost at the back of the ranks of pots and one in an 8 inch pot of compost at the front where the pot side was exposed to more sun. The pots did not contain plants and were occasionally watered (if only by the rain).
Download data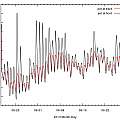
It can be see that the pot at the front got hotter than the one at the back. However in the warmest Summer for six years in England for pots of this size the high temperatures reached were not disastrous.
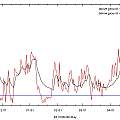
White pots are cooler than black
One solution to pots getting too hot that is often suggested is to use white pots. To push this idea to the limit, two brown 5 inch pots were filled with compost and put in a greenhouse, one of them was wrapped in aluminium foil.
Download data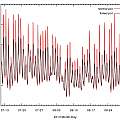
Yet again the common wisdom is shown to be correct. The reflective pot is cooler than the ordinary brown one. Temperatures reached in this experiment are higher than in the previous one, probably as a result of the pots being in a greenhouse and being smaller. Notice in the last two experiments, the pots that get hottest also get the coolest.
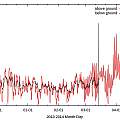
Recording temperatures for Winter 2013/2014
Temperatures are recorded using RC-3 temperature loggers. These record up to around 16,000 temperatures with a period that can be set from seconds to hours. They can be connected by a cable to a USB port for programming and data transfer. The devices are put in zip seal bags, which sit on a small plastic spacer in a lock tight food container, which is then tied in a plastic bag. The photos show an 8 inch pot, the temperature logger and packaging, the two loggers packaged, one logger in the pot, an 8 inch deep hole in to which the other logger was placed, and finally the 8 inch pot with logger next to a stick marking where the second one is buried. The time between measurements was set to one hour.
The site for the measurements is 500 feet above sea level in the North of England. The process in 2012/2013 was identical but the logger above ground was only in its lock tight box and not in a pot.
The graph below shows the results; it was a mild winter, temperatures only touched 0 °C on a handful of nights; the graph shows average temperatures in the pot higher than outside, and average temperatures underground higher still. It is expected that average temperatures will be more extreme in pots than in the ground and true temperatures be more volatile; in the context of Winter that means bulbs in the ground will be warmer than those in pots. In Summer pots are expected to be hotter than the ground. The underground temperature logger stopped recording on the 14th of March (shown by a spike to 20 °C) and the best lesson from this years data may be to set off with fresh batteries.
Download data
The Physics of container temperatures
The sky is cold You're stood on the promenade on a fine day in Summer, the sun shines down from a clear blue sky, looking out to sea you enjoy an ice cream. The hottest thing in this image is the sun, but what is the coldest? It is the blue sky.
Thermodynamics Heat moves from warm to cold objects (and not the other way around), as a result everything tends to end up the same temperature.
Radiation All objects radiate electromagnetic energy, if they're hot enough you can see it (think red hot pokers). They also absorb radiation. If the energy absorbed is greater than that radiated, they get hotter e.g. a pot sat in the sun. If the energy radiated is greater than that absorbed they cool down e.g. a pot outside on a clear night. Some objects do not absorb radiation well, they reflect it, for example anything white. Others absorb a lot, for example anything black. What is not so obvious is that poor absorbers are also poor radiators, and good absorbers are good radiators.
Conduction Heat moves between objects that are in contact with one another. A pot sat on the ground will tend to remain at the same temperature as the ground. Some materials are better conductors than others e.g water is more conductive than dry peat. The opposite of conduction is insulation.
Evaporation Water left open to the air disappears, it turns into water vapour in the air. Only the most energetic molecules have enough energy to do this, so evaporation cools down the remaining water. Plants 'transpire' taking water from the ground and evaporating it from their leaves. A pot of wet compost open to the air can cool itself by drying out.
Heat capacity In the way mass gives objects inertia (you have to push a massive object hard to move it), heat capacity gives them thermal inertia. An object with a large heat capacity needs a lot of energy adding to increase its temperature, or to lose a lot of energy to cool. A big pot of wet compost will suffer less variation in temperature than a small pot.